Are you getting the CBD you're paying for? What you need to know.
We all want to get exactly what we’re paying for. Unfortunately, all CBD products are not created equal. Unscrupulous companies make wild product claims or may mislabel or mislead about the amount of CBD in the product. Poor lab standards or manufacturing techniques can lead to a product that contains little or no CBD, or way too much. So how can you tell the quality CBDproducts from the rubbish?
What is CBD testing and why should I care?
In an industry that’s largely unregulated, many prominent companies have taken it upon themselves to self-impose quality control and testing standards. Most companies that take this step will be happy to share their Certificates of Analysis (CoA) with consumers. If an online manufacturer or a retail store doesn’t have the information, or refuses to share it, it is best to avoid that product and the retailer.
At Trove, we are proud of what goes into our CBD hemp products. So we offer full transparency of our analytics process. We test every batch of our CBD for potency, purity, and quality. This ensures our final products do not contain any detectable amount of THC. Additionally, we screen for the presence of any undesirable cannabinoids, bacteria, yeast, mold, residual solvents (VOC), pesticides, and heavy metals. Dose accuracy is vital to the efficacy of any health supplement. We perform regular lot testing on our entire product line to verify cannabinoid content and concentration so that you know exactly what you are buying.
To take this process a step further and close the gap between manufacturer and consumer, Trove places a label with a QR code on the bottom of every CBD product they make. This code can be scanned with any smart phone and will link directly to Trove’s website where we’ve made the CoA from every batch we’ve produced available to the public.
Why is this important? Due to the nature of the industry boom that has taken place with CBD, it’s hard to know who the legitimate manufacturers are. And without packaging and labeling standards, it may be difficult to know how much, if any, CBD is really in your product. Furthermore, the product you want to purchase may contain trace amounts of THC, which is still a controlled substance. It is important to know that your products are safe and legal. Confused about CoA's or how to read one? Read on.
How to read a Certificate of Analysis for CBD Products

Lab reports, known as Certificates of Analysis (CoA's), can be confusing to read for the uninitiated. So, let’s break these reports into their three main components:
- Sample Information
- Test Information
- Final Approval
Sample Information

The Sample Information section provides detail about the sample, the type of product submitted, and the test method used.
Sample Name – This is the name of the sample from the manufacturer. It should correspond to a product name.
Batch ID – The identifier provided by the manufacturer for internal tracking or traceability through the supply chain. It should correspond to the same Batch ID on your product or box.
Reported – The date the Certificate of Analysis was reported to the manufacturer.
Type – The Type refers to the matrix of the sample that was submitted. The categories are: plant, concentrate, solution, or unit.
Test – This field will list the type of test that was performed, e.g. potency, microbial, etc.
Test ID – The Test ID is a unique identifier for the sample and a requirement for ISO 17025.
Method – Each testing method has a number that links to an SOP. For example, “TM14” is a Cannabinoid Potency method.
Test Information
This is the bulk of the information on a report. The second section of the Certificate of Analysis will contain the data for the type of test that was performed. There are several types of tests that most reputable manufacturers will run, including: Cannabinoid Potency, Residual Solvent Analysis, Microbial Contamination, and Heavy Metals. But for the end consumer, Cannabinoid Potency is the most important, so that’s what we’ll focus on. This is the test that confirms how much CBD is in your product and if there are any other potentially unwanted cannabinoids present, such as THC. Legally, THC must be below 0.3% by weight to qualify as a hemp/CBD product. Beyond that threshold, it’s a cannabis product and therefore a controlled substance.
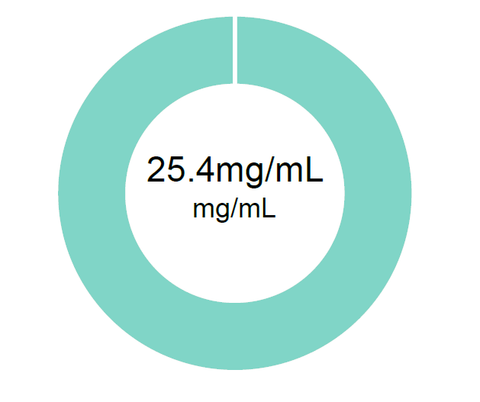
Donut Chart – This chart displays several data points. The donut ring will display each cannabinoid reported. Inside the donut is either the total cannabinoids reported in a percentage or for finished products, it will report the total milligrams of CBD.

Bar Chart – The bar chart is mainly for compliance purposes. It presents delta-9 THC, THCA, CBD, and CBDA.
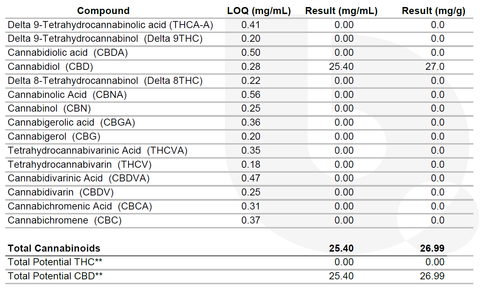
LOQ – LOQ means, “Limit of Quantitation”. This is the lowest level that the lab can accurately quantitate (or count) for each analyte in the report. The LOQ varies from sample to sample based on the weight and the dilution factor used for testing.
Result (mg/g) – This column displays the analyte reported in a milligram per gram amount.
Result (mg/mL) – When provided the density of the sample, the lab can display results in milligrams per milliliter.
Total Cannabinoids – The total cannabinoids section will display two values, Total “Potential” THC, and Total “Potential” CBD. This contains the theoretical conversion of THCA into delta-9 THC and CBDA into CBD.
Final Approval
The third section of a Certificate of Analysis is the Final Approval.

Prepared by/Date – The signature and name of the analyst that performed the test.
Approved by/Date – The signature and name of the quality assurance analyst that reviewed the test data.
Summary
There should always be a phone number for the laboratory on the report. If you as a consumer are concerned or need help understanding the lab report, you can always call the independent laboratory to verify the results. Or call the manufacturer’s customer service number, and they should be able to help you read and understand the report.
Still have questions? Call us at 1-833-GO-TROVE.
Copyright © 2020 Samuel Carter All rights reserved
